What Is Pharmaceutical Competitive Intelligence?
Pharmaceutical competitive intelligence is the disciplined practice of gathering, interpreting, and applying insights about competitors, market dynamics, regulatory developments, and stakeholder behavior to inform strategic decision-making across the drug development lifecycle.
But in reality, it is more than that.
In the pharma industry, where development cycles stretch over a decade and commercial outcomes are often determined in a matter of months, competitive intelligence functions as both an early-warning system and a strategic navigation tool. It helps organizations anticipate competitor moves before they materialize, pressure-test strategic assumptions, and identify opportunities to differentiate long before launch.
Too often, competitive intelligence is mistaken for simple monitoring. Tracking press releases, clinical trial updates, or conference abstracts is necessary, but it is not sufficient. True pharmaceutical competitive intelligence transforms information into implications. It connects dots across clinical development, commercial strategy, market access, and regulatory positioning to shape decisions that meaningfully impact performance.
Download the Complete Guide to Pharmaceutical Competitive Intelligence:
Pharma Competitive Intelligence vs Pharma Market Research
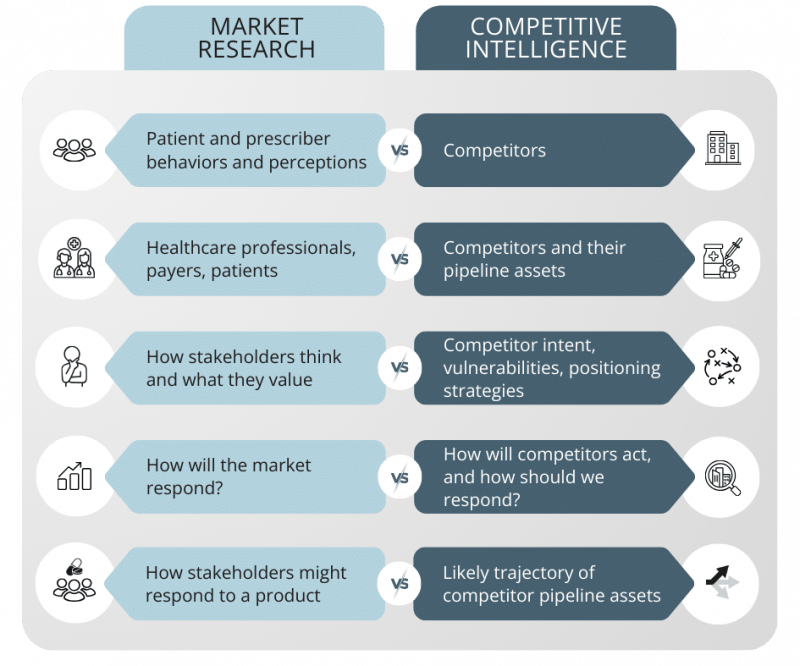
Competitive intelligence is frequently conflated with market research. While the two disciplines are complementary, they answer different strategic questions.
Market research primarily focuses on understanding patient and prescriber behaviors and perceptions. It explores how healthcare professionals, payers, and patients think, what they value, and how they might respond to a product.
Competitive intelligence, by contrast, focuses on competitors – their intent, their vulnerabilities, their positioning strategies, and the likely trajectory of their pipeline assets. Where market research asks, “How will the market respond?” competitive intelligence asks, “How will our competitors act, and how should we respond?”
High-performing pharmaceutical organizations integrate both disciplines. They use market research to refine engagement strategies and competitive intelligence to shape strategic positioning and resource allocation.
For a deeper look at how insights are translated into strategic implications rather than left as static reports, see framework for turning competitive intelligence into strategic implications.
The Scope of Pharmaceutical Competitive Intelligence
A comprehensive pharmaceutical competitive intelligence function spans multiple domains. It may involve monitoring competitor pipelines, analyzing clinical trial designs, tracking regulatory milestones, evaluating messaging shifts, and conducting primary intelligence interviews with key stakeholders.
However, the defining feature of strong CI is not the breadth of inputs, it is the depth of interpretation.
Effective competitive intelligence connects disparate signals into a coherent view of the competitive landscape. It identifies patterns in trial design, reads between the lines of investor communications, and anticipates how market access dynamics may evolve. It asks not only what competitors are doing today, but why, and what they are likely to do next.
For a broader overview of how CI integrates into launch execution and commercial strategy, see pharmaceutical competitive intelligence best practices and competitive intelligence for pharmaceutical launch planning.
Why Competitive Intelligence Is Critical in Today’s Pharmaceutical Landscape
The pharmaceutical landscape has become structurally more complex. As a result, the margin for strategic miscalculation has narrowed.
Crowded Pipelines and Compressed Differentiation
Many therapeutic areas that were once sparsely populated now feature multiple companies pursuing similar mechanisms of action. Oncology, immunology, neurology, and rare diseases routinely see parallel development programs racing toward similar endpoints.
In this environment, incremental improvements in efficacy may not be enough to secure meaningful market share. Differentiation must be strategically engineered.
Competitive intelligence allows organizations to understand where competitors are vulnerable, where they are strong, and where whitespace may exist. It informs trial design decisions, evidence generation strategies, and ultimately, positioning.
For broader context on evolving industry dynamics, see emerging pharma market trends shaping competition.
Escalating Market Access and Pricing Pressure
Commercial success is increasingly shaped by payer dynamics and access negotiations. Preferred formulary placement, rebate strategies, and health technology assessment outcomes can determine uptake curves as much as clinical performance.
Without incorporating competitive access strategy into forecasting, organizations risk overestimating early adoption and underestimating friction.
Competitive intelligence provides insight into how competitors are approaching pricing negotiations, how they are positioning value narratives, and where access barriers may emerge. This intelligence is especially critical during the early commercialization period, often referred to as the decisive first six months after approval.
For a detailed discussion of why early commercial execution is so critical, see the critical six-month pharma launch window.
Accelerated Competitive Milestones
The pace of competitor data releases, regulatory submissions, and label expansions has accelerated. A single Phase 3 data release can reshape prescribing behavior, alter payer negotiations, or compress differentiation windows.
Organizations must be prepared not just to observe competitor milestones, but to interpret them in real time and adjust strategy accordingly.
This requires disciplined milestone monitoring and structured response planning. Companies that lack this capability often find themselves reacting to shifts rather than anticipating them.
For more on proactive monitoring approaches, see how to track competitor regulatory and clinical milestones.
The Shrinking Launch Window
Perhaps the most consequential structural shift is the compression of the launch window. Increasingly, commercial trajectories are determined within months of approval. If rapid uptake is not achieved early, recovery becomes difficult.
Launch success is rarely won at approval. It is shaped years earlier through differentiated evidence generation, competitive positioning, and scenario planning.
Organizations that integrate competitive intelligence early in development are better positioned to enter the market with clarity and confidence. Those that defer CI engagement often struggle with reactive adjustments and misaligned expectations.
For an exploration of the common strategic errors that undermine launch success, see Common Pharma Launch Pitfalls.
The Role of Competitive Intelligence Across the Drug Development Lifecycle
One of the most persistent misconceptions about pharmaceutical competitive intelligence is that it is primarily a pre-launch activity. In reality, its strategic value begins in early development and continues well beyond commercialization.
High-performing pharmaceutical organizations align CI activities to each stage of the development lifecycle.
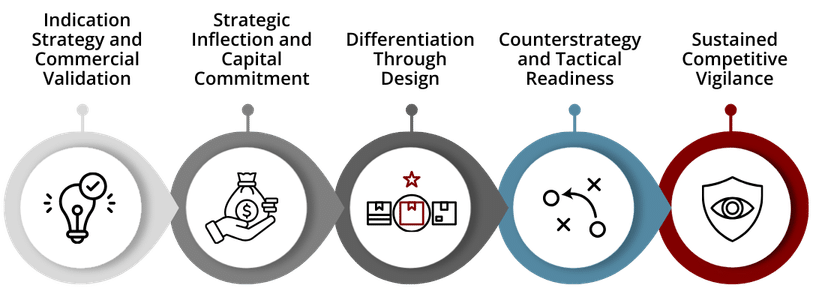
Early Development: Indication Strategy and Commercial Validation
In early development, competitive intelligence informs foundational decisions that can determine long-term viability.
At this stage, teams are assessing the size of the unmet need, evaluating competitive density, and determining whether a particular indication presents a defensible commercial opportunity. Misjudging the landscape at this point can result in years of development invested in a market that proves structurally unfavorable.
Competitive intelligence helps clarify:
- How crowded the mechanism space truly is
- Whether competitors are advancing faster than anticipated
- Where differentiation may realistically be achieved
For emerging biopharma companies in particular, early-stage competitive validation can prevent costly strategic missteps.
For a structured roadmap connecting early development to launch planning, see complete pharmaceutical launch planning guide.
Phase 2: Strategic Inflection and Capital Commitment
Phase 2 represents a pivotal moment. Proof-of-concept data begins to clarify potential, and organizations must decide whether to commit significant capital to Phase 3.
Competitive intelligence refines this decision by reassessing the landscape. It evaluates competitor timelines, monitors shifts in standard of care, and pressure-tests assumptions about differentiation.
At this stage, many organizations engage external CI partners to ensure objectivity and depth of analysis. Fresh perspective can help challenge internal optimism and validate strategic direction.
For guidance on evaluating strategic CI partners, see choosing a pharmaceutical CI partner.
Phase 3: Differentiation Through Design
In Phase 3, competitive intelligence becomes tightly linked to trial design and evidence generation.
Organizations benchmark competitor endpoints, evaluate patient population strategies, and assess how forthcoming data might be perceived by regulators and prescribers. Insights gathered at this stage can inform adjustments to evidence strategy, messaging preparation, and positioning.
Phase 3 is also when counter-strategy planning begins in earnest. Companies must consider how competitors will respond to their data, and how they will respond in turn.
For a comprehensive discussion of CI integration into launch readiness, see insights-driven pharma launch strategy.
Pre-Launch: Counterstrategy and Tactical Readiness
Approximately two years before anticipated approval, competitive intelligence activity intensifies. Monitoring becomes more granular, and strategic discussions shift toward tactical readiness.
Organizations analyze competitor messaging, field force deployment patterns, payer engagement approaches, and historical launch behavior. Scenario planning workshops are often conducted to stress-test potential responses to competitor moves.
Rather than assuming a static landscape, teams model possible outcomes and prepare contingency strategies.
For further exploration of this process, see competitive scenario planning in pharma and pharma field force competitive benchmarking analysis.
Post-Launch: Sustained Competitive Vigilance
Competitive intelligence does not conclude at approval. If anything, its importance increases.
After launch, competitors may expand indications, adjust pricing, introduce new data, or shift positioning. Ongoing monitoring ensures organizations can adapt lifecycle strategies accordingly.
Effective post-launch CI supports decisions related to resource allocation, evidence generation, label expansion, and portfolio prioritization.
Core Components of an Effective Pharmaceutical Competitive Intelligence Program
If pharmaceutical competitive intelligence is to function as a strategic discipline rather than a reporting function, it must be built deliberately.
An effective CI program is not defined by the volume of data collected, but by the clarity of implications generated. High-performing organizations move beyond passive monitoring and build structured systems that continuously inform strategic decisions.
While CI programs vary depending on company size and therapeutic focus, several foundational components consistently distinguish mature, high-impact functions.
Structured Competitor Monitoring
At its most basic level, competitive intelligence begins with systematic monitoring. However, structured monitoring is far more sophisticated than tracking headlines.
It involves defining which competitors matter most, which milestones are most consequential, and which signals require escalation. Monitoring must be tailored to the stage of development and commercial maturity of the asset.
In early development, emphasis may be placed on pipeline movements and mechanism-of-action shifts. In Phase 3 and pre-launch stages, attention shifts toward trial design nuances, regulatory signals, and messaging preparation.
The goal is not to gather more data. It is to identify the signals that could meaningfully alter strategy.
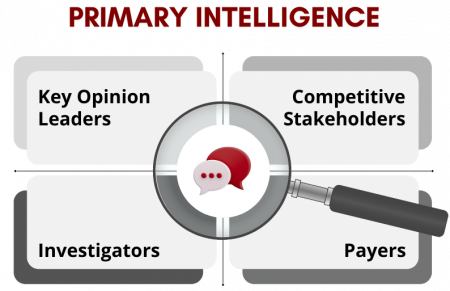
Primary Intelligence and Hard-to-Reach Sources
Secondary research – press releases, earnings calls, conference abstracts – provides a baseline understanding of the competitive landscape. But by definition, this information is publicly available to everyone.
Primary intelligence differentiates leading CI programs. Through structured interviews with key opinion leaders, investigators, payers, and occasionally competitor stakeholders, organizations gain insight into perceptions, adoption drivers, and strategic intent that are not visible in the public domain.
Primary intelligence helps answer questions such as:
- How is competitor data being interpreted in clinical practice?
- Are payers viewing a new asset as incremental or transformative?
- Are trial endpoints perceived as meaningful or marginal?
- How is a competitor’s messaging, positioning, and commercial model evolving?
These nuanced perspectives often reveal gaps between how companies position their products and how the market actually receives them.
For more on this approach, see Pharmaceutical Primary Intelligence.
Competitive Scenario Planning
Static planning is insufficient. Effective CI programs incorporate structured scenario planning to prepare for multiple possible outcomes.
Rather than assuming a single data scenario or regulatory pathway, leading teams model “if-then” situations. If a competitor receives accelerated approval, how does that alter positioning? If Phase 3 data underperforms expectations, how should resource allocation shift? If pricing pressure intensifies, which segments remain defensible?
Scenario workshops bring together clinical, commercial, and market access teams to align around potential responses before events occur.
This disciplined preparation reduces reactive decision-making and increases strategic agility.
For more on this structured approach, see Pharma Scenario Planning.
Commercial and Field Intelligence
As products approach launch, the competitive battlefield extends beyond clinical data. Messaging, field force deployment, promotional intensity, and relationship strength begin to influence market share.
Understanding how competitors structure their field organizations, which customer segments they prioritize, and how they frame their value propositions can significantly influence tactical planning.
Field intelligence often reveals competitive advantages or vulnerabilities that are not visible in trial data alone. It also informs resource allocation decisions and training priorities for commercial teams.
For further discussion, see Pharma Field Force Analysis.
Translating Intelligence into Strategic Implications
Perhaps the most critical, and most frequently overlooked, component of an effective CI function is disciplined implication development.
Data alone does not drive strategic impact. Interpretation does.
High-performing CI teams consistently translate intelligence into clear strategic questions:
- What does this mean for our positioning?
- How should this influence our evidence generation plan?
- Does this alter our forecast assumptions?
- Are we adequately differentiated in the eyes of stakeholders?
This shift from reporting to interpretation elevates CI from a support function to a strategic partner.
For a structured framework on turning intelligence into implications, see Pharma Insights and Implications.
Common Pitfalls in Pharmaceutical Competitive Intelligence
Despite the growing recognition of its importance, many pharmaceutical organizations struggle to extract full value from competitive intelligence.
The most common pitfalls do not stem from lack of effort, they stem from structural misalignment.
Treating CI as a Reporting Function
One of the most pervasive issues is reducing competitive intelligence to static reporting. Weekly updates, slide decks, and milestone summaries are produced, but their implications are rarely integrated into decision-making processes.
When CI is confined to information dissemination rather than strategic dialogue, it loses influence. Reports may be read, but strategy remains unchanged.
Organizations that derive meaningful impact from CI ensure it has direct access to decision-makers and participates in cross-functional planning discussions.
Over-Reliance on Secondary Research
Publicly available information provides important context, but it rarely provides competitive advantage.
Companies that rely exclusively on secondary sources risk missing critical nuance. They may understand what competitors are saying, but not how the market is interpreting it.
Integrating primary intelligence into CI programs provides a deeper layer of insight and reduces the risk of strategic blind spots.
Engaging Competitive Intelligence Too Late
Another common error is deferring competitive intelligence investment until the final stages of development.
By the time Phase 3 trials are underway, many strategic decisions, including indication focus, endpoint selection, and evidence generation, have already been locked in. At that point, CI may highlight vulnerabilities, but the ability to course-correct is limited.
Organizations that begin structured competitive monitoring in Phase 2, or even earlier, are better positioned to shape differentiation proactively rather than reactively.
Failure to Integrate Cross-Functionally
Competitive intelligence operates at the intersection of clinical development, commercial strategy, regulatory affairs, and market access. When CI is siloed within a single function — particularly if embedded too narrowly — its perspective becomes constrained.
Cross-functional integration ensures that intelligence informs trial design, pricing strategy, forecasting models, and promotional messaging in a coordinated way.
Organizations that achieve higher levels of launch success tend to treat CI as a connective tissue rather than a peripheral function.
Underestimating the Dynamic Nature of Competitors
Perhaps the most subtle pitfall is assuming competitors will remain static.
Competitors refine messaging, adjust pricing strategies, modify trial designs, and accelerate timelines. They respond to market signals just as actively as your own organization.
CI must therefore operate as a continuous discipline. Monitoring cannot pause after approval, nor can scenario planning stop once a strategy is defined.
Together, these pitfalls highlight a central theme: pharmaceutical competitive intelligence delivers value only when it is embedded early, interpreted rigorously, and integrated broadly.
How High-Performing Pharmaceutical Organizations Structure Competitive Intelligence
The effectiveness of pharmaceutical competitive intelligence is not determined solely by tools or resources. It is shaped by how the function is structured, positioned, and integrated within the organization.
In many companies, CI begins as an embedded support role, often sitting within commercial, business development, or strategy. While this structure can work in smaller organizations, it can also limit influence if the function is too narrowly aligned with one stakeholder group.
High-performing pharmaceutical organizations tend to share several structural characteristics.
Clear Strategic Mandate
Competitive intelligence functions that exert meaningful influence typically have a clearly defined mandate: to inform strategic decision-making across the development and commercialization lifecycle.
This mandate extends beyond reporting competitor activity. It includes participating in portfolio reviews, launch planning discussions, forecasting refinement, and cross-functional scenario planning.
When CI is formally recognized as a strategic partner rather than a reactive service provider, its insights carry greater weight.
Cross-Functional Access
CI derives its strength from perspective. To maintain that perspective, it must have visibility across clinical development, medical affairs, regulatory strategy, commercial operations, and market access.
Organizations that restrict CI to one function risk narrowing its lens. By contrast, those that encourage cross-functional engagement allow intelligence to influence decisions more holistically.
Frequent engagement with stakeholders — whether through structured updates, working sessions, or scenario workshops — increases both strategic influence and stakeholder trust.
Integration Across the Development Timeline
High-performing CI functions are active long before launch.
They support early indication strategy, inform Phase 2 investment decisions, contribute to trial design refinement, and intensify monitoring during the pre-launch period. After approval, they continue tracking competitor lifecycle management strategies and emerging threats.
Organizations that limit CI engagement to the final stages of development often find that key differentiation decisions have already been made.
Balanced Use of Internal and External Resources
Even large pharmaceutical companies rarely maintain sufficient internal bandwidth to conduct deep primary intelligence, attend every relevant congress, and run structured scenario workshops independently.
High-performing organizations often supplement internal CI teams with external partners at key inflection points, particularly during Phase 2 validation, Phase 3 differentiation planning, and pre-launch counter-strategy modeling.
This hybrid model combines internal institutional knowledge with external objectivity and specialized expertise.
Ultimately, structure matters because it determines influence. When CI is embedded early, integrated broadly, and supported adequately, it evolves from a monitoring function into a strategic advantage.
When Should Pharmaceutical Companies Engage External Competitive Intelligence Services?
The question is not whether pharmaceutical companies should engage external competitive intelligence services. The more relevant question is when and why.
While some organizations attempt to build entirely internal CI capabilities, development complexity and competitive acceleration often create inflection points where external pharma competitive intelligence services become strategically valuable.
Early Development: Market Opportunity Validation
In preclinical and early Phase 1 stages, companies are still defining indication focus and evaluating commercial viability.
At this stage, an objective external perspective can challenge internal assumptions about unmet need, competitive density, and differentiation potential. This is particularly important for emerging biopharma organizations, where a misjudged opportunity can have existential consequences.
External pharma competitive intelligence firms can provide independent landscape assessments and primary intelligence that validates, or refines, early strategic direction.
For a structured overview of early-stage launch planning considerations, see Pharmaceutical Launch Planning Guide.
Phase 2: Capital Commitment and Strategic Risk
Phase 2 is often the most consequential inflection point in development. Organizations must decide whether to commit significant capital to Phase 3.
Engaging external CI services at this stage provides:
- Objective reassessment of the competitive landscape
- Deeper primary intelligence validation
- Structured risk modeling
- Independent scenario planning
External partners can also help pressure-test internal optimism and ensure that differentiation is both meaningful and defensible.
Phase 3 and Pre-Launch: Counter-Strategy and Tactical Preparation
As Phase 3 data emerges and approval approaches, competitive intensity increases.
This is often when organizations engage external CI partners for:
- Competitive simulation workshops
- Milestone monitoring acceleration
- Field force benchmarking
- Payer intelligence refinement
- Launch readiness stress testing
External objectivity is particularly valuable during this period, as internal teams may become deeply invested in optimistic positioning narratives.
Post-Launch: Sustaining Advantage
External CI support can also play a role after approval, particularly in monitoring competitor lifecycle expansions, emerging biosimilars, pricing shifts, and evolving evidence narratives.
Sustained monitoring helps prevent complacency and ensures that lifecycle management decisions are grounded in competitive reality.
What to Look for in a Pharmaceutical Competitive Intelligence Partner
Not all CI providers deliver the same value. When evaluating external support, pharmaceutical organizations should consider several critical capabilities.
- First, access to high-quality primary intelligence is essential. Publicly available data can be collected internally. True differentiation comes from insight that cannot be easily replicated.
- Second, therapeutic expertise matters. Understanding disease-specific dynamics, regulatory nuances, and standard-of-care evolution enhances interpretation accuracy.
- Third, structured strategic frameworks ensure that intelligence is translated into implications rather than presented as disconnected findings.
- Finally, ethical standards must be uncompromising. Intelligence gathering must remain compliant, transparent, and aligned with industry regulations.
Engaging external pharmaceutical competitive intelligence services should not be viewed as a sign of internal deficiency. Rather, it is often a strategic decision to enhance objectivity, expand capability, and strengthen launch confidence.
The Future of Pharmaceutical Competitive Intelligence
Pharmaceutical competitive intelligence is entering a new era.
The volume of available data has expanded dramatically. Clinical trial registries update in real time. Conference abstracts are released digitally. Regulatory signals are increasingly transparent. Social media and digital engagement add new layers of competitive noise.
At the same time, artificial intelligence and automation tools are accelerating information processing. Large language models can summarize trial results within seconds. Monitoring platforms can flag competitor press releases instantly.
But while access to information has increased, the ability to interpret it strategically remains the defining advantage.
The future of pharmaceutical competitive intelligence will not be determined by who gathers the most data. It will be determined by who translates that data into strategic foresight most effectively.
Several trends are likely to shape the evolution of CI in the coming years.
Greater Integration of AI, With Human Oversight
Automation will continue to enhance monitoring efficiency. AI can accelerate document review, trend identification, and signal detection.
However, competitive intelligence in pharma involves nuance. It requires understanding therapeutic context, regulatory history, stakeholder psychology, and commercial behavior. These elements cannot be reduced to pattern recognition alone.
The highest-performing CI models will combine AI-enabled efficiency with human interpretation. Technology will surface signals. Experienced analysts will interpret implications.
Earlier Lifecycle Engagement
Competitive intelligence will increasingly shift upstream in the development timeline. Organizations will begin structured CI engagement in preclinical and Phase 1 stages rather than waiting until pre-launch.
As differentiation windows narrow, proactive strategy becomes more valuable than reactive adjustment.
Stronger Cross-Functional Alignment
CI will continue evolving from a reporting function into a connective function. It will sit at the intersection of clinical development, regulatory strategy, medical affairs, commercial planning, and market access.
Increased Demand for Objectivity
As competitive intensity rises, internal bias becomes more dangerous. Optimism bias, confirmation bias, and sunk-cost bias can distort interpretation of competitor signals.
External perspectives, whether through advisory input or structured competitive simulations, will play a larger role in preserving strategic objectivity.
Key Takeaways: Building a Strategic Pharmaceutical Competitive Intelligence Function
Across the drug development lifecycle, several principles consistently differentiate high-performing organizations:
- Competitive intelligence must begin early, ideally by Phase 2 or sooner.
- Monitoring alone is insufficient, interpretation and implication development drive impact.
- Cross-functional integration increases strategic influence.
- Scenario planning reduces reactive decision-making.
- Primary intelligence provides differentiation beyond public information.
- Sustained post-launch vigilance preserves market position.
Organizations that treat CI as a lifecycle discipline, rather than a pre-launch exercise, are better equipped to navigate crowded pipelines, pricing pressure, regulatory acceleration, and compressed commercialization windows.
The companies that outperform are rarely those with the most data. They are those with the clearest strategic interpretation of it.
Strengthen Your Pharmaceutical Competitive Intelligence Strategy
At Sedulo Group, we partner with pharmaceutical and biotech organizations to transform competitive intelligence into strategic advantage.
Our approach integrates:
- Structured competitor monitoring across the lifecycle
- Deep primary intelligence from hard-to-reach sources
- Competitive simulation and scenario planning workshops
- Field force and commercial intelligence benchmarking
- Strategic implication development grounded in cross-functional alignment
From early-stage indication validation through launch execution and lifecycle management, we help organizations move from reactive monitoring to proactive strategy.
If you are evaluating how to strengthen your pharmaceutical competitive intelligence capabilities, or preparing for a critical development or launch inflection point, we welcome the opportunity to discuss your strategic priorities.
Explore Sedulo’s pharmaceutical competitive intelligence services or contact our team to begin a conversation about how intelligence can drive launch excellence and long-term competitive advantage.
Frequently Asked Questions About Pharmaceutical Competitive Intelligence
What is pharmaceutical competitive intelligence?
Pharmaceutical competitive intelligence is the structured process of gathering, analyzing, and interpreting information about competitors, clinical developments, regulatory activity, and market dynamics to inform strategic decision-making across the drug development lifecycle. It helps companies anticipate competitor actions and refine differentiation strategies before and after launch.
What is the difference between competitive intelligence and market research in pharma?
Market research focuses on understanding customer perceptions and behaviors among prescribers, payers, and patients. Competitive intelligence focuses on competitors, their strategy, positioning, vulnerabilities, and likely next moves. Both disciplines are complementary but serve different strategic purposes.
When should a pharmaceutical company engage competitive intelligence services?
Pharmaceutical companies often engage competitive intelligence services during key inflection points, including Phase 2 validation, Phase 3 differentiation planning, pre-launch preparation, and post-launch lifecycle management. Early engagement allows organizations to proactively shape strategy rather than react to competitor milestones.
What are pharmaceutical competitive intelligence services?
Pharmaceutical competitive intelligence services typically include competitor monitoring, milestone tracking, primary intelligence interviews, scenario planning workshops, launch simulations, field force analysis, and strategic implication development. These services support clinical development, commercialization, and lifecycle strategy.
How is competitive intelligence used during drug launch?
During drug launch, competitive intelligence informs positioning, messaging strategy, payer negotiation preparation, field force deployment, and counter-strategy modeling. It helps teams anticipate competitor responses and protect early uptake during the critical first six months after approval.
How do you measure the impact of competitive intelligence?
Competitive intelligence impact can be measured by its influence on strategic decisions, forecasting accuracy, launch readiness preparedness, risk mitigation, and stakeholder satisfaction. High-performing organizations evaluate CI based on how it shapes decisions, not simply how much information it produces.

